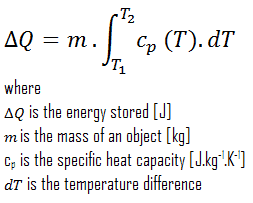Thermal energy storage forms a key component of a power plant to improve its dispatchability, especially for concentrating solar power plants (CSP). Thermal energy storage (TES) is achieved with widely differing technologies. There are three methods used and still being investigated to store thermal energy.
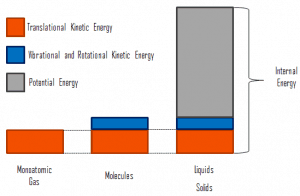 In thermodynamics, internal energy (also called thermal energy) is defined as the energy associated with microscopic forms of energy. It is an extensive quantity, and it depends on the size of the system or on the amount of substance it contains. The SI unit of internal energy is the joule (J). It is the energy contained within the system, excluding the kinetic energy of motion of the system as a whole and the system’s potential energy. Microscopic forms of energy include those due to the rotation, vibration, translation, and interactions among the molecules of a substance. None of these forms of energy can be measured or evaluated directly. Still, techniques have been developed to evaluate the change in the total sum of all these microscopic forms of energy.
In thermodynamics, internal energy (also called thermal energy) is defined as the energy associated with microscopic forms of energy. It is an extensive quantity, and it depends on the size of the system or on the amount of substance it contains. The SI unit of internal energy is the joule (J). It is the energy contained within the system, excluding the kinetic energy of motion of the system as a whole and the system’s potential energy. Microscopic forms of energy include those due to the rotation, vibration, translation, and interactions among the molecules of a substance. None of these forms of energy can be measured or evaluated directly. Still, techniques have been developed to evaluate the change in the total sum of all these microscopic forms of energy.
In addition, energy can be stored in the chemical bonds between the atoms that make up the molecules. This energy storage on the atomic level includes energy associated with electron orbital states, nuclear spin, and binding forces in the nucleus.

Thermal energy can also be very effectively stored. Nowadays, the situation in energy markets is different. The increase in the prices of conventional energy sources and environmental awareness has led to an increase in the use of renewable energies and energy efficiency. Thermal energy storage forms a key component of a power plant to improve its dispatchability, especially for concentrating solar power plants (CSP). Thermal energy storage (TES) is achieved with widely differing technologies. There are three methods used and still being investigated to store thermal energy.
Sensible Heat Storage (SHS)
The most direct way is the storage of sensible heat. Sensible heat storage is based on raising the temperature of a liquid or solid to store heat and releasing it with a decrease in temperature when required. The volumes needed to store energy on the scale that the world needs are extremely large. Materials used in sensible heat storage must have a high heat capacity and high boiling or melting point. Although this method of heat storage is currently less efficient for heat storage, it is least complicated compared with latent or chemical heat, and it is inexpensive.
From a thermodynamics point of view, the storage of sensible heat is based on the increase of enthalpy of the material in the store, either a liquid or a solid, in most cases. The sensible effect is a temperature change. Heat stored can be obtained by the equation:
Heat Capacity
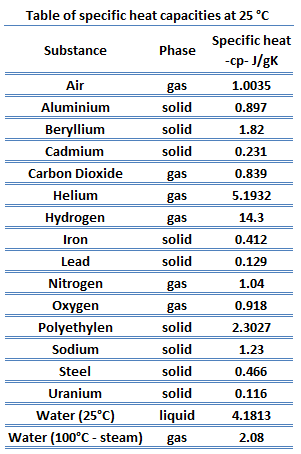 Different substances are affected to different magnitudes by the addition of heat. When a given amount of heat is added to different substances, their temperatures increase by different amounts. This proportionality constant between the heat Q that the object absorbs or loses and the resulting temperature change T of the object is known as the heat capacity C of an object.
Different substances are affected to different magnitudes by the addition of heat. When a given amount of heat is added to different substances, their temperatures increase by different amounts. This proportionality constant between the heat Q that the object absorbs or loses and the resulting temperature change T of the object is known as the heat capacity C of an object.
C = Q / ΔT
Heat capacity is an extensive property of matter, meaning it is proportional to the size of the system. Heat capacity C has the unit of energy per degree or energy per kelvin. When expressing the same phenomenon as an intensive property, the heat capacity is divided by the amount of substance, mass, or volume. Thus the quantity is independent of the size or extent of the sample.
Latent Heat Storage (LHS)
A common approach to thermal energy storage is to use materials known as phase change materials (PCMs). These materials store heat when they undergo a phase change, for example, from solid to liquid, from liquid to gas, or from solid to solid (change of one crystalline form into another without a physical phase change).
The phase change “solid-to-liquid” is the most used, but solid-to-solid change is of interest. These materials can be used as an effective way of storing thermal energy (solar energy, off-peak electricity, industrial waste heat). In comparison to sensible heat storage systems, latent heat storage has the advantages of high storage density (due to high latent heat of fusion) and the isothermal nature of the storage process. The heat of fusion or the heat of evaporation is much greater than the specific heat capacity. The comparison between latent heat storage and sensible heat storage shows that latent heat storage densities are typically 5 to 10 times higher.
In general, latent heat effects associated with the phase change are significant. Latent heat, also known as the enthalpy of vaporization (liquid-to-vapor phase change) or enthalpy of fusion (solid-to-liquid phase change), is the amount of heat added to or removed from a substance to produce a phase change. This energy breaks down the intermolecular attractive forces and must provide the energy necessary to expand the substance (the pΔV work). When latent heat is added, no temperature change occurs.
Phase Change Material
Phase Change Materials (PCM) are latent heat storage materials. It is possible to find materials with a latent heat of fusion and melting temperature inside the desired range. The PCM to be used in the design of thermal storage systems should accomplish desirable thermophysical, kinetics, and chemical properties.
Thermo-physical Properties
- Suitable phase-transition temperature for the specific application.
- High latent heat of phase transition to occupy the minimum possible volume.
- Melting temperature in the desired operating temperature range.
- High specific heat to provide for additional significant sensible heat storage.
- High thermal conductivity to minimize temperature gradient and assist the charging and discharging of energy of the storage systems.
- Small volume changes on phase transformation and small vapor pressure at operating temperatures reduce the containment problem.
Kinetic Properties
- High nucleation rate to avoid supercooling of the liquid phase.
- High rate of crystal growth so that the system can meet demands of heat recovery from the storage system.
Chemical Properties
- Non-toxic, non-flammable, and non-explosive materials for safety reasons.
- Long-term chemical stability and complete reversible melt/freeze cycle.
- No degradation after a large number of freeze/melt cycles.
- Low corrosivity
Finally, the material must be abundant, available, and cheap to help in the feasibility of using the storage system.
There are a large number of PCMs, and they can be divided into three groups:
- Organic PCMs
- Inorganic PCMs
- Eutectic PCMs
As an example, thermal energy storage can concentrate solar power stations (CSP). The principal advantage is the ability to efficiently store energy, allowing the dispatching of electricity over up to a 24-hour period. In a CSP plant that includes storage, the solar energy is first used to heat the molten salt or synthetic oil to store thermal energy at high temperatures in insulated tanks. Later hot molten salt is used for steam production to generate electricity by steam turbo generator as per requirement. Using latent heat and sensible heat in concentrating solar power stations is possible with high-temperature solar thermal input. Various eutectic mixtures of metals, such as Aluminium and Silicon (AlSi12), offer a high melting point (577°C) suited to efficient steam generation. In contrast, high alumina cement-based materials offer good thermal storage capabilities.
Thermo-chemical Storage
One of three possible approaches to thermal energy storage is reversible thermo-chemical reactions. The most important advantage of the thermo-chemical storage method is that the enthalpy of reaction is considerably larger than the specific heat or the heat of fusion. Therefore the storage density is much better. In chemical reactions, energy is stored in the chemical bonds between the atoms that make up the molecules. Energy storage on the atomic level includes energy associated with electron orbital states. Whether a chemical reaction absorbs or releases energy, there is no overall change in the amount of energy during the reaction. That’s because of the law of conservation of energy, which states that:
Energy cannot be created or destroyed. Energy may change form during a chemical reaction.
One example of an experimental storage system based on chemical reaction energy is salt hydrate technology. The system is especially advantageous for seasonal thermal energy storage. The system uses the reaction energy created when salts are hydrated or dehydrated. It works by storing heat in a container containing 50% sodium hydroxide (NaOH) solution. Heat (e.g., using a solar collector) is stored by evaporating the water in an endothermic reaction. When water is added again, heat is released in an exothermic reaction at 50 °C. Current systems operate at 60% efficiency.