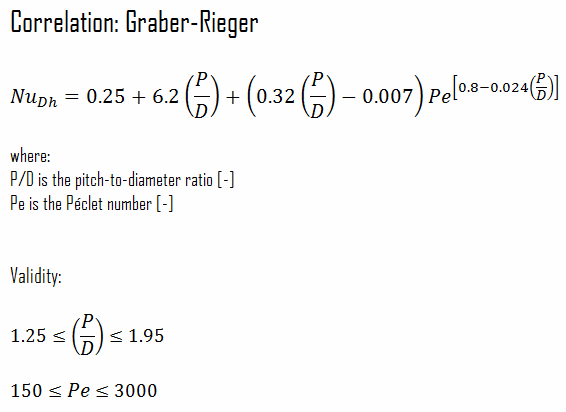In physics, liquid metal consists of alloy with very low melting points, which form a eutectic that is liquid at room temperature. In reactor engineering, liquid metals are alloys with low melting points allowing for reactor coolant to be liquid in operating range of temperatures (usually above the room temperature).
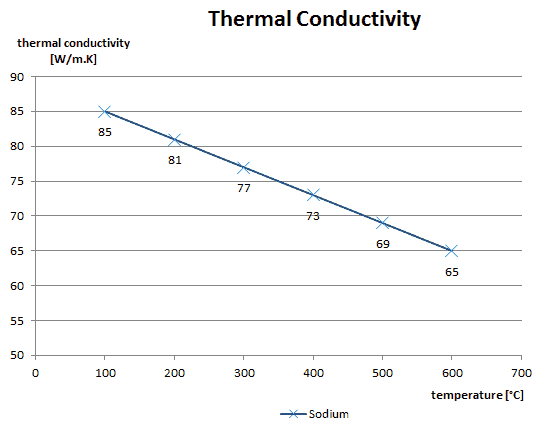
Liquid metals can be used as a reactor coolant because they have excellent heat transfer properties and can be employed in low-pressure systems, as is the case of sodium-cooled fast reactors (SFRs). The unique feature of metals as far as their structure is concerned is the presence of charge carriers, specifically free electrons, giving them high electrical conductivity high thermal conductivity.
The use of liquid metal coolants made it possible to provide a high rate of heat transfer in power plants as well as the temperatures of working surfaces of their constructions close to coolant temperature.
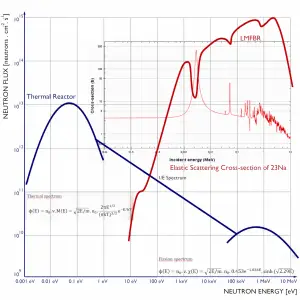
Moreover, liquid metals used in reactor engineering are weak absorbers of neutrons, allowing liquid metal reactors to operate with a fast neutron spectrum. A liquid metal fast reactor is a high power density reactor, which does not need a neutron moderator.
The main differences between thermal and fast reactors are, of course, in neutron cross-sections that exhibit significant energy dependency. It can be characterized by the capture-to-fission ratio, which is lower in fast reactors. There is also a difference in the number of neutrons produced per one fission, which is higher in fast reactors than in thermal reactors. These very important differences are caused primarily by differences in neutron fluxes, and therefore it is very important to know detailed neutron energy distribution in a reactor core.
The disadvantage of many liquid metals is their high chemical activity at interaction with oxygen, water, and structural materials, which may cause heat transfer deterioration in the plant under certain conditions.
Sodium and NaK – Reactor Coolant
Sodium-cooled fast reactors (SFRs) are the most common fast reactor design. They use molten sodium or a eutectic sodium-potassium alloy (NaK) as the reactor coolant. Melting and boiling points of sodium and NaK are:
-
Source: wikipedia.org License: Public domain sodium
- melting point – 97.72°C
- boiling point – 883°C
- NaK – a eutectic mixture
- melting point – (-12°C)
- boiling point – 785°C
Source: wikipedia.org
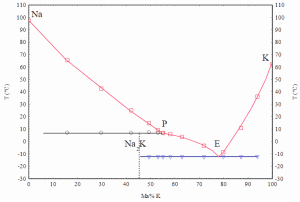
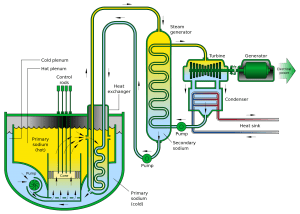
NaK containing 40% to 90% potassium by weight is liquid at room temperature. The eutectic mixture consists of 77% potassium and 23% sodium. Sodium and NaK do not corrode steel significantly and are compatible with many nuclear fuels, allowing for a wide choice of structural materials. Because sodium reacts violently with water, however, SFRs require the placement of an intermediate heat exchanger between the reactor core and the steam generator. This hi-tech technology requires a lot of experience. Therefore, only a few countries have developed their fast reactor design.
Lead and Lead-bismuth Eutectic – Reactor Coolant
Lead, lead-bismuth eutectic, and other metals have been proposed and occasionally used. The lead-cooled fast reactor is a nuclear reactor design that features a fast neutron spectrum and molten lead or lead-bismuth eutectic coolant. Lead-Bismuth Eutectic or LBE is a eutectic alloy of lead (44.5%) and bismuth (55.5%). Molten lead or lead-bismuth eutectic is the primary coolant because lead and bismuth have low neutron absorption and relatively low melting points.
Melting and boiling points of lead and lead-bismuth eutectic mixture are:
- lead
- melting point – 327.5°C
- boiling point – 1749°C
- lead-bismuth – a eutectic mixture
- melting point – 123.5°C
- boiling point – 1670°C
Source: wikipedia.org
Compared to sodium-based liquid metal coolants such as liquid sodium or NaK, lead-based coolants have significantly higher boiling points, meaning a reactor can be operated without the risk of coolant boiling at much higher temperatures. Lead, and LBE does not react readily with water or air, in contrast to sodium and NaK, which ignite spontaneously in air and react explosively with water. Due to its denseness and high atomic number, lead and bismuth are excellent gamma radiation shields while simultaneously virtually transparent to neutrons.
On the other hand, lead, and LBE coolants are more corrosive to steel than sodium or NaK eutectic alloy. This and the very high density of lead puts an upper limit on the velocity of coolant flow through the reactor due to safety considerations. Furthermore, the higher melting points of lead and LBE (327 °C and 123.5 °C, respectively) may mean that the solidification of the coolant may be a greater problem when the reactor is operated at lower temperatures.
Nusselt Number for Liquid Metal Reactors
A liquid metal cooled reactor is an advanced type of nuclear reactor where the primary coolant is a liquid metal. Liquid metals can be used as a coolant because they have excellent heat transfer properties and can be employed in low-pressure systems, as is the case of sodium-cooled fast reactors (SFRs). The unique feature of metals as far as their structure is concerned is the presence of charge carriers, specifically free electrons, giving them high electrical conductivity high thermal conductivity. This very high thermal conductivity, together with low viscosity causes, that typical heat transfer correlations (e.g., Dittus-Boelter) can not be used.
Heat transfer coefficients for sodium flow through the fuel channel are based on the Prandtl number and Péclet number. Pitch-to-diameter (P/D) also enters many calculations of heat transfer in liquid metal reactors. Convective heat transfer correlations are usually presented in terms of Nusselt number versus Péclet number. Typical Péclet numbers for normal operation are from 150 to 300 in the fuel bundles. As for other flow regimes, the Nusselt number and a given correlation can be used to determine the convective heat transfer coefficient.
- Sodium-cooled fast reactor
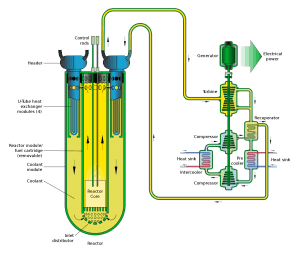
- Lead-cooled fast reactor
One of the main challenges in numerical simulation is the reliable modeling of heat transfer in liquid-metal cooled reactors by Computational Fluid Dynamics (CFD). Heat transfer applications with low-Prandtl number fluids are often in the transition range between conduction and convection dominated regimes.