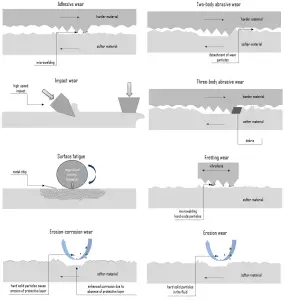
 In general, wear is mechanically induced surface damage that results in the progressive removal of material due to relative motion between that surface and a contacting substance or substances. A contacting substance may consist of another surface, a fluid, or hard, abrasive particles contained in some form of fluid or suspension, such as a lubricant. As is with friction, the presence of wear can be either good or bad. Productive, controlled wear can be found in processes like machining, cutting, grinding, and polishing. However, in most technological applications, the occurrence of wear is highly undesirable and is an enormously expensive problem since it leads to the deterioration or failure of components. In terms of safety, it is often not as serious (or as sudden) as a fracture, and this is because the wear is usually anticipated.
In general, wear is mechanically induced surface damage that results in the progressive removal of material due to relative motion between that surface and a contacting substance or substances. A contacting substance may consist of another surface, a fluid, or hard, abrasive particles contained in some form of fluid or suspension, such as a lubricant. As is with friction, the presence of wear can be either good or bad. Productive, controlled wear can be found in processes like machining, cutting, grinding, and polishing. However, in most technological applications, the occurrence of wear is highly undesirable and is an enormously expensive problem since it leads to the deterioration or failure of components. In terms of safety, it is often not as serious (or as sudden) as a fracture, and this is because the wear is usually anticipated.
Certain material characteristics such as hardness, carbide type, and volume percent can have a decided impact on the wear resistance of a material in a given application. Wear, like corrosion, has multiple types and subtypes that are predictable to some extent and are rather difficult to test and evaluate in the lab or service reliably.
Diffusive wear
Diffusion or dissolution wear refers to the damage, erosion, or degradation of materials on a metal’s surface due to increased surface temperatures. When two materials are in contact, atoms from one material could diffuse into the other, causing diffusion or dissolution wear. Diffusive wear is primarily due to the heat produced by adhesion when two rough surfaces move across each other, typically when one metal is sliding across the other.