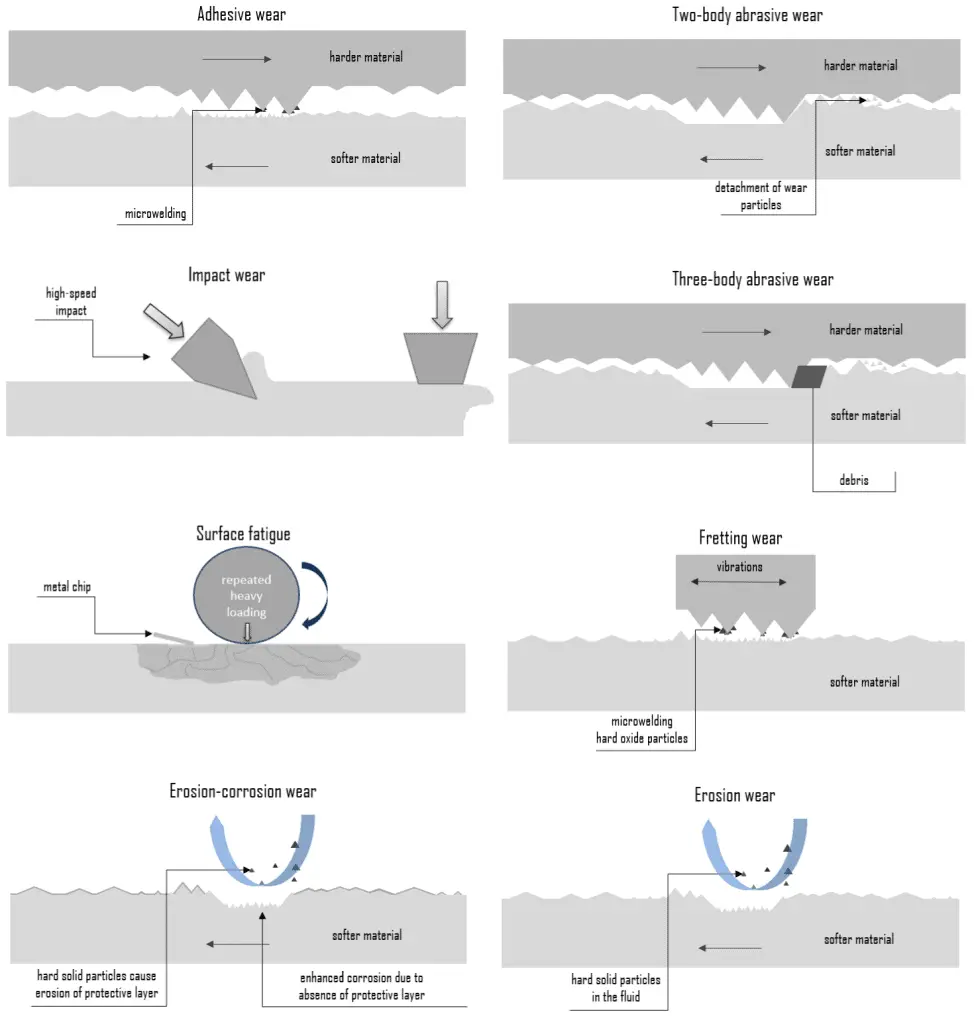
 In general, wear is mechanically induced surface damage that results in the progressive removal of material due to relative motion between that surface and a contacting substance or substances. A contacting substance may consist of another surface, a fluid, or hard, abrasive particles contained in some form of fluid or suspension, such as a lubricant. As is with friction, the presence of wear can be either good or bad. Productive, controlled wear can be found in processes like machining, cutting, grinding, and polishing. However, in most technological applications, the occurrence of wear is highly undesirable, and it is an enormously expensive problem since it leads to the deterioration or failure of components. In terms of safety, it is often not as serious (or as sudden) as a fracture, and this is because the wear is usually anticipated.
In general, wear is mechanically induced surface damage that results in the progressive removal of material due to relative motion between that surface and a contacting substance or substances. A contacting substance may consist of another surface, a fluid, or hard, abrasive particles contained in some form of fluid or suspension, such as a lubricant. As is with friction, the presence of wear can be either good or bad. Productive, controlled wear can be found in processes like machining, cutting, grinding, and polishing. However, in most technological applications, the occurrence of wear is highly undesirable, and it is an enormously expensive problem since it leads to the deterioration or failure of components. In terms of safety, it is often not as serious (or as sudden) as a fracture, and this is because the wear is usually anticipated.
Certain material characteristics such as hardness, carbide type, and volume percent can have a decided impact on the wear resistance of a material in a given application. Wear, like corrosion, has multiple types and subtypes that are predictable to some extent and are rather difficult to test reliably and evaluate in the lab or service.
Surface Hardness and Wear Resistance
Hardness is important from an engineering standpoint because resistance to wear by either friction or erosion by steam, oil, and water generally increases with hardness. If the hardness of the material is higher than that of the abrasive material, less wear rate will occur.
Case hardening or surface hardening is the process in which the hardness of an object’s surface (case) is enhanced while the inner core of the object remains elastic and tough. After this process enhances surface hardness, wear resistance, and fatigue life. This is accomplished by several processes, such as a carburizing or nitriding process by which a component is exposed to a carbonaceous or nitrogenous atmosphere at elevated temperatures. As was written, two main material characteristics are influenced:
- Hardness and wear resistance is significantly enhanced. In materials science, hardness is the ability to withstand surface indentation (localized plastic deformation) and scratching. Hardness is probably the most poorly defined material property because it may indicate resistance to scratching, abrasion, indentation, or even resistance to shaping or localized plastic deformation. Hardness is important from an engineering standpoint because resistance to wear by either friction or erosion by steam, oil, and water generally increases with hardness.
- Toughness is not negatively influenced, and toughness is the ability of a material to absorb energy and plastically deform without fracturing. One definition of toughness (for high-strain rate, fracture toughness) is that it is a property that is indicative of a material’s resistance to fracture when a crack (or other stress-concentrating defects) is present.
The case-hardening process involves infusing additional carbon or nitrogen into the surface layer for iron or steel with low carbon content, which has poor to no hardenability. Case hardening is useful in parts such as a cam or ring gear that must have a very hard surface to resist wear and a tough interior to resist the impact that occurs during operation. Further, the surface hardening of steel has an advantage over hardening (that is, hardening the metal uniformly throughout the piece) because less expensive low-carbon and medium-carbon steels can be surface hardened without the problems of distortion and cracking associated with the through hardening of thick sections. A carbon- or nitrogen-rich outer surface layer (or case) is introduced by atomic diffusion from the gaseous phase. The case is normally 1 mm deep and is harder than the inner core of the material.