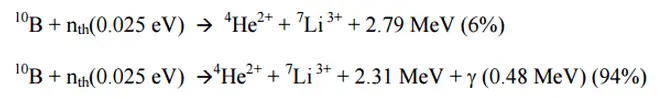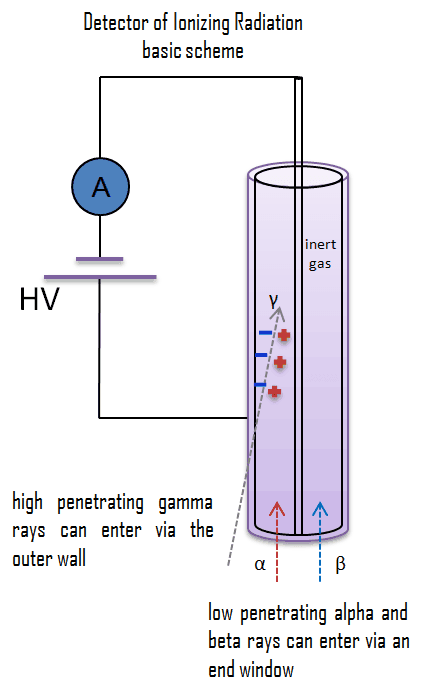
The ionization chamber, also known as the ion chamber, is an electrical device that detects various types of ionizing radiation. The voltage of the detector is adjusted so that the conditions correspond to the ionization region, and the voltage is insufficient to produce gas amplification (secondary ionization). Detectors in the ionization region operate at a low electric field strength, so no gas multiplication occurs. The charge collected (output signal) is independent of the applied voltage. Single minimum-ionizing particles tend to be quite small and usually require special low-noise amplifiers for efficient operating performance. Ionization chambers are preferred for high radiation dose rates because they have no “dead time,” a phenomenon that affects the accuracy of the Geiger-Mueller tube at high dose rates. This is because there is no inherent amplification of signal in the operating medium; therefore, these counters do not require much time to recover from large currents. In addition, because there is no amplification, they provide excellent energy resolution, which is limited primarily by electronic noise.
Ionization chambers can be operated in current or pulse mode. In contrast, pulse mode almost always uses proportional counters or Geiger counters. Detectors of ionizing radiation can be used both for activity measurements as well as for dose measurements. The dose can be obtained with knowledge about the energy needed to form a pair of ions.
Detection of Alpha Radiation using Ionization Chamber
For alpha and beta particles to be detected by ionization chambers, they must be given a thin window. This “end-window” must be thin enough for the alpha and beta particles to penetrate. However, a window of almost any thickness will prevent an alpha particle from entering the chamber. The window is usually made of mica with a density of about 1.5 – 2.0 mg/cm2. But it does not mean alpha radiation cannot be detected by an ionization chamber.
For example, in some kinds of smoke detectors, you can meet artificial radionuclides such as americium-241, a source of alpha particles. The smoke detector has two ionization chambers, one open to the air and a reference chamber that does not allow the entry of particles. The radioactive source emits alpha particles into both chambers, which ionizes some air molecules. The free-air chamber allows the entry of smoke particles into the sensitive volume and changes the attenuation of alpha particles. If any smoke particles enter the free-air chamber, some ions will attach to the particles and not be available to carry the current in that chamber. An electronic circuit detects that a current difference has developed between the open and sealed chambers and sounds the alarm.
Detection of Beta Radiation using Ionization Chamber
For alpha and beta particles to be detected by ionization chambers, they must be given a thin window. This “end-window” must be thin enough for the alpha and beta particles to penetrate. However, a window of almost any thickness will prevent an alpha particle from entering the chamber. The window is usually made of mica with a density of about 1.5 – 2.0 mg/cm2.
An ionization chamber may be, for example, used for the measurement of tritium in the air. These devices are known as tritium-in-air monitors. Tritium is a radioactive isotope but emits a very weak form of radiation, a low-energy beta particle similar to an electron. It is a pure beta emitter (i.e., beta emitter without accompanying gamma radiation). The electron’s kinetic energy varies, with an average of 5.7 keV, while the remaining energy is carried off by the nearly undetectable electron antineutrino. Such a very low energy of electron causes that the electron cannot penetrate the skin or even does not travel very far in the air. Beta particles from tritium can penetrate only about 6.0 mm of air. Designing a detector whose walls these beta particles can penetrate is practically impossible. Instead, the tritium-in-air monitor pumps the tritium-contaminated air right through an ionization chamber so that all of the energy of the beta particles can be usefully converted to producing ion pairs inside the chamber.
Detection of Gamma Radiation using Ionization Chamber
Gamma rays have very little trouble penetrating the metal walls of the chamber. Therefore, ionization chambers may be used to detect gamma radiation and X-rays, collectively known as photons, and for this, the windowless tube is used. Ionization chambers have a uniform response to radiation over a wide range of energies and are the preferred means of measuring high levels of gamma radiation. Some problems are caused by alpha particles being more ionizing than beta particles, and gamma rays, so more current is produced in the ionization chamber region by alpha than beta and gamma. Gamma rays deposit a significantly lower amount of energy into the detector than other particles.
The efficiency of the chamber can be further increased by the use of high-pressure gas. Typically a pressure of 8-10 atmospheres can be used, and various noble gases are employed. For example, high-pressure xenon (HPXe) ionization chambers are ideal for use in uncontrolled environments, as a detector’s response is uniform over large temperature ranges (20–170°C). The higher pressure results in a greater gas density and thereby a greater chance of collision with the fill gas and ion-pair creation by incident gamma radiation. Because of the increased wall thickness required to withstand this high pressure, only gamma radiation can be detected. These detectors are used in survey meters and for environmental monitoring.
Detection of Neutrons using Ionization Chamber
Since the neutrons are electrically neutral particles, they are mainly subject to strong nuclear forces but not to electric forces. Therefore, neutrons are not directly ionizing and usually convert into charged particles before they can be detected. Generally, every type of neutron detector must be equipped with a converter (to convert neutron radiation to common detectable radiation) and one of the conventional radiation detectors (scintillation detector, gaseous detector, semiconductor detector, etc.).
Ionization chambers are often used as the charged particle detection device. For example, if the inner surface of the ionization chamber is coated with a thin coat of boron, the (n, alpha) reaction can occur. ost of (n,alpha) reactions of thermal neutrons are 10B(n,alpha)7Li reactions accompanied by 0.48 MeV gamma emission.
Moreover, isotope boron-10 has a high (n, alpha) reaction cross-section along the entire neutron energy spectrum. The alpha particle causes ionization within the chamber, and ejected electrons cause further secondary ionizations.
Another method for detecting neutrons using an ionization chamber is to use the gas boron trifluoride (BF3) instead of air in the chamber. The incoming neutrons produce alpha particles when reacting with the boron atoms in the detector gas. Either method may be used to detect neutrons in a nuclear reactor. It must be noted that BF3 counters are usually operated in the proportional region.