Thallium-doped Sodium Iodide – NaI(Tl) scintillators
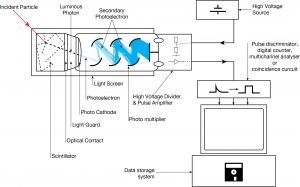
NaI(Tl) (thallium-doped sodium iodide) is the most widely used scintillation material. NaI(Tl) as the scintillator is used in scintillation detectors, traditionally in nuclear medicine, geophysics, nuclear physics, and environmental measurements. The iodine provides most of the stopping power in sodium iodide (since it has a high Z = 53). These crystalline scintillators are characterized by high density, high atomic number, and pulse decay times of approximately 1 microsecond (~ 10-6 sec). The wavelength of maximum emission is 415 nm. Scintillation in inorganic crystals is typically slower than in organic ones. They exhibit high efficiency for the detection of gamma rays and are capable of handling high count rates. Inorganic crystals can be cut to small sizes and arranged in an array configuration to provide position sensitivity. This feature is widely used in medical imaging to detect X-rays or gamma rays. Inorganic scintillators are better at detecting gamma rays and X-rays. This is due to their high density and atomic number, which gives a high electron density. A disadvantage of some inorganic crystals, e.g., NaI, is their hygroscopicity, a property that requires them to be housed in an airtight container to protect them from moisture. The crystals are usually coupled with a photomultiplier tube in a hermetically sealed assembly.