 Radiation is all around us. In, around, and above the world we live in. It is a natural energy force that surrounds us, and it is a part of our natural world that has been here since the birth of our planet. From the beginning of time, all living creatures have been, and are still being, exposed to ionizing radiation. Ionizing radiation is generated through nuclear reactions, nuclear decay, very high temperature, or acceleration of charged particles in electromagnetic fields. This radiation is known as background radiation. Background radiation is ionizing radiation present in the environment that originates from various natural and artificial sources.
Radiation is all around us. In, around, and above the world we live in. It is a natural energy force that surrounds us, and it is a part of our natural world that has been here since the birth of our planet. From the beginning of time, all living creatures have been, and are still being, exposed to ionizing radiation. Ionizing radiation is generated through nuclear reactions, nuclear decay, very high temperature, or acceleration of charged particles in electromagnetic fields. This radiation is known as background radiation. Background radiation is ionizing radiation present in the environment that originates from various natural and artificial sources.
There are two broad categories of radiation sources:
- Natural Background Radiation. Natural background radiation includes radiation produced by the Sun, lightning, primordial radioisotopes, supernova explosions, etc.
- Man-Made Sources of Radiation. Artificial sources include medical uses of radiation, residues from nuclear tests, industrial uses of radiation, etc.
Special Reference: Sources and effects of ionizing radiation, Annex B. UNSCEAR. New York, 2010. ISBN: 978-92-1-142274-0.
Natural Background Radiation
Natural background radiation is ionizing radiation that originates from various natural sources. From the beginning of time, all living creatures have been, and are still being, exposed to ionizing radiation. This radiation is not associated with any human activity. There are radioactive isotopes in our bodies, houses, air, water, and soil. We all are also exposed to radiation from outer space.
Sources of Natural Background Radiation
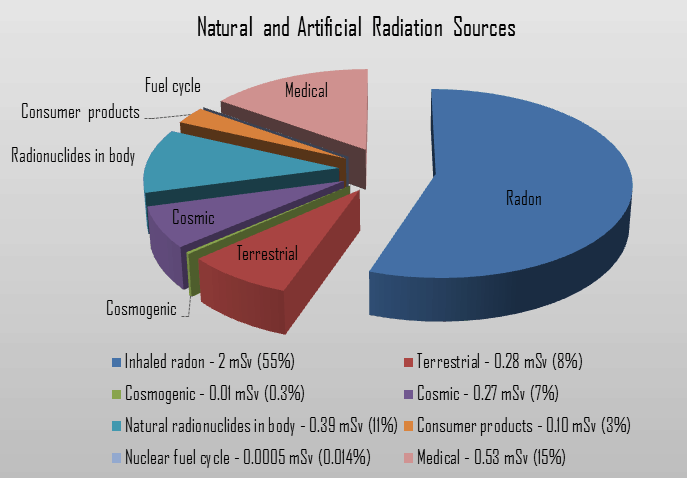
We divide all these natural radiation sources into three groups:
- Cosmic Radiation. Cosmic radiation refers to sources of radiation from cosmic rays that come from the Sun or outer space. At ground level, the muons, with energies mostly between 1 and 20 GeV, contribute about 75 % of the absorbed dose rate in free air. The remainder comes from electrons produced by the muons or present in the electromagnetic cascade. The annual cosmic ray dose at sea level is around 0.27 mSv (27 mrem). If you live at higher elevations or are a frequent airline passenger, this exposure can be significantly higher since the atmosphere is thinner here. The effects of the earth’s magnetic field also determine the dose from cosmic radiation.
- Terrestrial Radiation. Terrestrial radiation refers to radiation sources in the soil, water, and vegetation. The major isotopes of concern for terrestrial radiation are uranium and the decay products of uranium, such as thorium, radium, and radon. The average dose rate that originates from terrestrial nuclides (except radon exposure) is about 0.057 µGy/hr. The maximum values have been measured on monazite sand in Guarapari, Brazil (up to 50 µGy/hr and in Kerala, India (about 2 µGy/hr), and on rocks with a high radium concentration in Ramsar, Iran (from 1 to 10 µGy/hr). The average annual radiation dose to a person from radon is about 2 mSv/year, which may vary over many orders of magnitude from place to place. Radon is so important that it is usually treated separately.
- Internal Radiation. In addition to the cosmic and terrestrial sources, all people also have radioactive potassium-40, carbon-14, lead-210, and other isotopes inside their bodies from birth. The concentration of potassium-40 is nearly stable in all persons at a level of about 55 Bq/kg (3850 Bq in total), which corresponds to the annual effective dose of 0.2 mSv. The annual dose from carbon-14 is estimated to be about 12 μSv/year.
Man-made Sources of Radiation
Since ionizing radiation has many industrial and medical uses, people can also be exposed to artificial sources of radiation. Artificial sources include medical uses of radiation, residues from nuclear tests, industrial uses of radiation, television, and numerous other radiation-producing devices. For example, in some kinds of smoke detectors, you can meet artificial radionuclides such as americium-241. This artificial radionuclide is used to ionize the air and detect smoke.
It must be noted that most of these exposures are very low in intensity and the total dose and do not pose larger health effects. In each case, the usefulness of ionizing radiation must be balanced with its hazards. Nowadays, a compromise has been found, and most of the uses of radiation are optimized. Today it is almost unbelievable that x-rays were, at one time, used to find the right pair of shoes (i.e., shoe-fitting fluoroscopy). Measurements made in recent years indicate that the doses to the feet were in the range of 0.07 – 0.14 Gy for a 20-second exposure. This practice was halted when the risks of ionizing radiation were better understood.
In general, the following artificial sources expose the public to radiation:
- Medical Exposures (by far, the most significant artificial source)
- Diagnostic x-rays
- Nuclear medicine procedures (iodine-131, cesium-137, technetium-99m etc.)
- Consumer Products
- Building and road construction materials
- Smoking cigarettes (polonium-210)
- Combustible fuels, including gas and coal
- X-ray security systems
- Televisions
- Smoke detectors (americium)
- Lantern mantles (thorium)
To a lesser degree, the public is also exposed to radiation from the nuclear fuel cycle, from uranium mining and milling to the disposal of used (spent) fuel. The public is also exposed to radiation from so-called “enhanced sources of naturally occurring radioactive material.” This also means industries such as metal mining, coal mining, and power production from coal create additional exposures due to the densification of naturally occurring radionuclides. The public receives minimal exposure from the transportation of radioactive materials and fallout from nuclear weapons testing and reactor accidents (such as Chernobyl).
Background Radiation and Health Hazard
You can not go through life without radiation. The danger of ionizing radiation lies in the fact that the radiation is invisible and not directly detectable by human senses. People can neither see nor feel radiation, yet it deposits energy into the molecules of the body.
But don’t worry. The doses from background radiation are usually very small (except for radon exposure). A low dose means additional small doses comparable to the normal background radiation (10 µSv = average daily dose received from natural background). The problem is that, at very low doses, it is practically impossible to correlate any irradiation with certain biological effects. This is because the baseline cancer rate is already very high, and the risk of developing cancer fluctuates by 40% because of individual lifestyle and environmental effects, obscuring the subtle effects of low-level radiation.
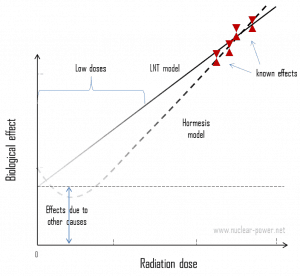
Secondly, and this is crucial, the truth about low-dose radiation health effects still needs to be found. It is unknown whether these low doses of radiation are detrimental or beneficial (and where is the threshold). Government and regulatory bodies assume an LNT model instead of a threshold or hormesis not because it is the more scientifically convincing but because it is, the more conservative estimate. The problem with this model is that it neglects many defense biological processes that may be crucial at low doses. The research during the last two decades is very interesting and shows that small doses of radiation given at a low dose rate stimulate the defense mechanisms. Therefore the LNT model is not universally accepted, with some proposing an adaptive dose-response relationship where low doses are protective, and high doses are detrimental. Many studies have contradicted the LNT model, and many have shown adaptive response to low dose radiation resulting in reduced mutations and cancers. This phenomenon is known as radiation hormesis.
According to the radiation hormesis hypothesis, radiation exposure comparable to and just above the natural background level of radiation is not harmful but beneficial, while accepting that much higher radiation levels are hazardous. Arguments for hormesis center around some large-scale epidemiological studies and the evidence from animal irradiation experiments, but most notably the recent advances in knowledge of the adaptive response. Proponents of radiation hormesis typically claim that radio-protective responses in cells and the immune system not only counter radiation’s harmful effects but also inhibit spontaneous cancer not related to radiation exposure.
See also: LNT Model