In addition to chemical and nonradioactive carcinogens, tobacco and tobacco smoke contain small amounts of lead-210 and polonium-210, both of which are radioactive carcinogens. It must be emphasized that cigarettes and tobacco also contain polonium-210, originating from the decay products of radon, which stick to tobacco leaves. Polonium-210 emits a 5.3 MeV alpha particle, which provides most of the equivalent dose. Due to decay of polonium-210, the annual local dose to localized spots at the bifurcations of segmental bronchi in the lungs caused by smoking cigarettes (1.5 packs/day) is about 80 mSv/year. Heavy smoking results in a dose of 160 mSv/year. This dose is not readily comparable to the radiation protection limits since the latter deal with whole-body doses, while the dose from smoking is delivered to a very small portion of the body. Many researchers believe that doses of polonium-210 are the origin of the high incidence of lung cancer among smokers.
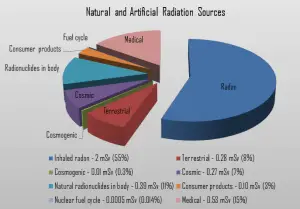
Recall that lead-210 and polonium-210 are daughter nuclei of radon-222. Radon-222 is a gas produced by the decay of radium-226. Both are a part of the natural uranium series. Since uranium is found in soil worldwide in varying concentrations, the dose of gaseous radon varies worldwide. Radon-222 is the most important and most stable isotope of radon. It has a half-life of only 3.8 days, making radon one of the rarest elements since it decays away quickly. An important source of natural radiation is radon gas, which seeps continuously from bedrock but can, because of its high density, accumulates in the ground. The fact radon is gas plays a crucial role in the spreading of all its daughter nuclei. As radon-222 decays into lead-210, lead-210 can be attached to dust of moisture particles and be stuck to tobacco leaves. When these particles are concentrated by smoking and inhaled as smoke, some of lead-210 is retained by the body. Since lead-210 is a weak beta emitter, it does not cause major doses, but polonium-210 does.
See also: Radon – Health Effects
The polonium-210, the decay product of lead-210, emits a 5.3 MeV alpha particle, which provides most of the equivalent dose. Alpha particles that belong to high-LET radiation are fairly massive and carry a double positive charge. They tend to travel only a short distance and do not penetrate very far into a tissue, if at all. However, alpha particles will deposit their energy over a smaller volume (possibly only a few cells if they enter a body) and cause more damage to those few cells (more than 80 % of the absorbed energy from radon is due to the alpha particles). Therefore, the radiation weighting factor for alpha radiation is equal to 20. An absorbed dose of 1 mGy by alpha particles will lead to an equivalent dose of 20 mSv.
Special Reference: Sources and effects of ionizing radiation, Annex B. UNSCEAR. New York, 2010. ISBN: 978-92-1-142274-0.