Radon-222 is a radioisotope of radon. Radon is a colorless, odorless, tasteless noble gas, occurring naturally as the decay product of radium. All isotopes of radon are radioactive, but the two radon isotopes, radon-222 and radon-220, are very important from a radiation protection point of view.
-
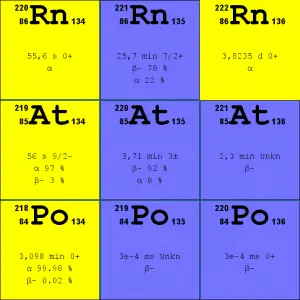
Source: JANIS (Java-based Nuclear Data Information Software); ENDF/B-VII.1 Radon-222. The radon-222 isotope is a natural decay product of the most stable uranium isotope (uranium-238). Thus it is a member of the uranium series.
- Radon-220. The radon-220 isotope, commonly referred to as thoron, is a natural decay product of the most stable thorium isotope (thorium-232). Thus it is a member of the thorium series.
It is important to note that radon is a noble gas, whereas all its decay products are metals. The main mechanism for radon entry into the atmosphere is diffusion through the soil. As a gas, radon diffuses through rocks and the soil. When radon disintegrates, the daughter metallic isotopes are ions that will be attached to other molecules like water and aerosol particles in the air. Therefore all discussions of radon concentrations in the environment refer to radon-222. While the average rate of production of radon-220 (thoron) is about the same as that of radon-222, the amount of radon-220 in the environment is much less than that of radon-222 because of the significantly shorter half-life (it has less time to diffuse) of radon-222 (55 seconds, versus 3.8 days respectively). Simply radon-220 has a lower chance of escaping from bedrock.
See also: Radon – Health Effects
Radon-222

Radon-222 is a gas produced by the decay of radium-226. Both are a part of the natural uranium series. Since uranium is found in soil worldwide in varying concentrations, the dose of gaseous radon varies worldwide. Radon-222 is the most important and most stable isotope of radon. It has a half-life of only 3.8 days, making radon one of the rarest elements since it decays away quickly. An important source of natural radiation is radon gas, which seeps continuously from bedrock but can accumulate in poorly ventilated houses because of its high density. The fact radon is gas plays a crucial role in the spreading of all its daughter nuclei. Simply radon is a transport medium from bedrock to the atmosphere (or inside buildings) for its short-lived decay products (Pb-210 and Po-210), which posses much more health risks.
Radon is usually the largest natural source of radiation contributing to the exposure of public members, sometimes accounting for half the total exposure (about 2 mSv/year) from all sources. The health risk due to exposure to radon and thoron comes principally from the inhalation of the short-lived decay products (Pb-210 and Po-210) and the resulting alpha-particle irradiation of the bronchi and the lungs.
As long as these isotopes are outside the body, only the gamma radiation will be able to give a dose. But radon is a gas that diffuses from the ground to mix with air. The half-life of radon-222 is long compared with the residence time of air in the lungs so that relatively little radon decay during respiration. Moreover, radon is a noble gas, and its inertness prevents its long-term retention within the body. But when radon disintegrates, the daughter metallic isotopes (Pb-210 and Po-210) are not inert and are attached to other molecules like water and aerosol particles in the air. When these particles are inhaled, some of lead-210 is retained by the body. Ingestion of lead-210 is also a possible way. Since lead-210 is a weak beta emitter, it does not cause major doses. Lead-210 is thus a transport medium from indoor air to the body. The radiation from radon and its decay products is a mixture of alpha particles, beta particles, and gamma radiation. When the isotopes come inside the body, all types of radiation contribute.
But it is the polonium-210, the decay product of lead-210, that emits a 5.3 MeV alpha particle, which provides most of the equivalent dose. Alpha particles that belong to high-LET radiation are fairly massive and carry a double positive charge. They tend to travel only a short distance and do not penetrate very far into a tissue, if at all. However, alpha particles will deposit their energy over a smaller volume (possibly only a few cells if they enter a body) and cause more damage to those few cells (more than 80 % of the absorbed energy from radon is due to the alpha particles). Therefore, the radiation weighting factor for alpha radiation is equal to 20. An absorbed dose of 1 mGy by alpha particles will lead to an equivalent dose of 20 mSv. In summary, radon and lead can be viewed as different sorts of carriers for polonium-210.
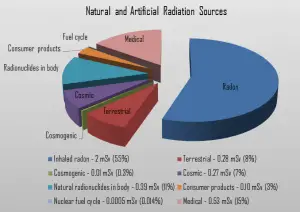 The amount of isotopes ingested with the food is negligible, and all concern is the breathing and the deposition of radon daughters in the bronchi and the lungs. Among non-smokers, radon is the largest cause of lung cancer and, overall, the second-leading cause. The average annual radiation dose to a person from radon is about 2 mSv/year, which may vary over many orders of magnitude from place to place. According to a 2003 report, EPA’s Assessment of Risks from Radon in Homes, epidemiological evidence shows a clear link between lung cancer and high radon concentrations.
The amount of isotopes ingested with the food is negligible, and all concern is the breathing and the deposition of radon daughters in the bronchi and the lungs. Among non-smokers, radon is the largest cause of lung cancer and, overall, the second-leading cause. The average annual radiation dose to a person from radon is about 2 mSv/year, which may vary over many orders of magnitude from place to place. According to a 2003 report, EPA’s Assessment of Risks from Radon in Homes, epidemiological evidence shows a clear link between lung cancer and high radon concentrations.
It must be emphasized that cigarettes also contain polonium-210, originating from the decay products of radon, which stick to tobacco leaves. Polonium-210 emits a 5.3 MeV alpha particle, which provides most of the equivalent dose. Heavy smoking results in a dose of 160 mSv/year to localized spots at the bifurcations of segmental bronchi in the lungs from the decay of polonium-210. This dose is not readily comparable to the radiation protection limits since the latter deal with whole-body doses. In contrast, the dose from smoking is delivered to a very small portion of the body.