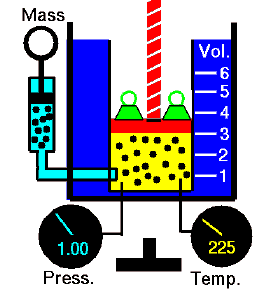
Isochoric Process – Isometric Process
An isochoric process is a thermodynamic process in which the volume of the closed system remains constant (V = const). It describes the behavior of gas inside the container that cannot be deformed. Since the volume remains constant, the heat transfer into or out of the system does not the p∆V work but only changes the system’s internal energy (the temperature).
In engineering internal combustion engines, isochoric processes are very important for their thermodynamic cycles (Otto and Diesel cycle). Therefore the study of this process is crucial for automotive engineering.
Example of Isochoric Process – Isochoric Heat Addition
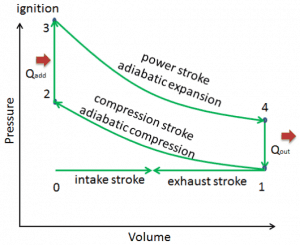 Let assume the Otto Cycle, which is one of the most common thermodynamic cycles found in automobile engines. This cycle assumes that the heat addition occurs instantaneously (between 2 → 3) while the piston is at the top dead center. This process is considered to be isochoric.
Let assume the Otto Cycle, which is one of the most common thermodynamic cycles found in automobile engines. This cycle assumes that the heat addition occurs instantaneously (between 2 → 3) while the piston is at the top dead center. This process is considered to be isochoric.
Processes 2 → 3, and 4 → 1 are isochoric processes. The heat is transferred into the system between 2 → 3 and out of the system between 4 → 1. During these processes, no work is done on the system or extracted from the system. The isochoric process 2 → 3 is intended to represent the ignition of the fuel-air mixture and the subsequent rapid burning.