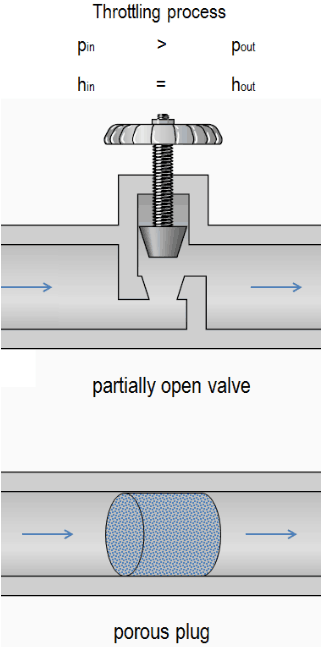
A throttling process is a thermodynamic process in which the enthalpy of the gas or medium remains constant (h = const). The throttling process is one of the isenthalpic processes. During the throttling process, no work is done by or on the system (dW = 0), and usually, there is no heat transfer (adiabatic) from or into the system (dQ = 0). On the other, the throttling process cannot be isentropic. It is a fundamentally irreversible process. Characteristics of throttling process:
- No Work Transfer
- No Heat Transfer
- Irreversible Process
- Isenthalpic Process
Example: Throttling of Wet Steam
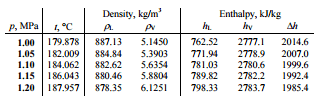
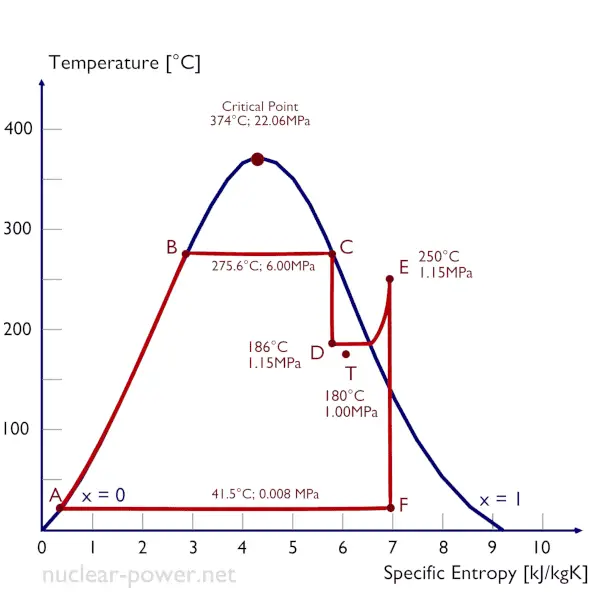
A high-pressure steam turbine stage operates at a steady state with inlet conditions of 6 MPa, t = 275.6°C, x = 1 (point C). Steam leaves this turbine stage at a pressure of 1.15 MPa, 186°C, and x = 0.87 (point D). Determine the vapor quality of the steam when throttled from 1.15 MPa to 1.0 MPa. Assume the process is adiabatic and no work is done by the system.
See also: Steam Tables
Solution:
The enthalpy for the state D must be calculated using vapor quality:
hD, wet = hD,vapor x + (1 – x ) hD,liquid = 2782 . 0.87 + (1 – 0.87) . 790 = 2420 + 103 = 2523 kJ/kg
Since it is an isenthalpic process, we know the enthalpy for point T. From steam tables we have to find the vapor quality using the same equation and solving the equation for vapor quality, x:
hT, wet = hT,vapor x + (1 – x ) hT,liquid
x = (hT, wet – hT, liquid) / (hT, vapor – hT, liquid) = (2523 – 762) / (2777 – 762) = 0.874 = 87.4%
In this case of the throttling process (1.15MPa to 1MPa) the vapor quality increases from 87% to 87.4% and the temperature decreases from 186°C to 179.9°C.