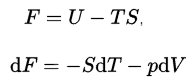In thermodynamics, the Helmholtz free energy is a thermodynamic potential defined as the internal energy of the system minus the product of the temperature times the entropy of the system. It measures the “useful” work obtainable from a closed thermodynamic system at a constant volume and pressure. The Helmhotz free energy is defined as:
The internal energy, U, has an exact physical meaning. It is the sum of all the kinetic and potential energies of all the particles in the system. The second term is the amount of spontaneous energy transfer, TS, where S is the final entropy of the system. For a constant temperature process, the Helmholtz free energy gives all the reversible work. When physicists say “free energy” without indicating Helmholtz or Gibbs, they usually mean Helmholtz free energy. On the other hand, when chemists say “free energy,” they almost always mean Gibbs free energy.