Thorium 232
Thorium 232, which alone makes up nearly all natural Thorium, is the most common isotope of Thorium in nature. This isotope has the longest half-life (1.4 x 1010 years) of all isotopes with more than 83 protons. Its half-life is considerably longer than the age of the Earth. Therefore 232Th belongs to primordial nuclides.232Th decays via alpha decay into 228Ra. 232Th occasionally decays by spontaneous fission with a very low probability of 1.1 x 10-9 %.
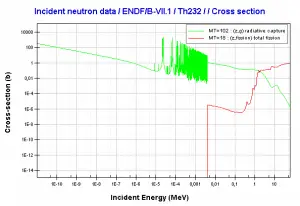
232Th is a fertile isotope. 232Th is not capable of undergoing a fission reaction after absorbing a thermal neutron. On the other hand, 232Th can be fissioned by fast neutron with energy higher than >1MeV. 232Th does not also meet the alternative requirement to fissile materials. 232Th cannot sustain a nuclear fission chain reaction because too many neutrons produced by fission of 232Th have lower energies than the original neutron. Isotope 232Th is the key material in the thorium fuel cycle. Radiative capture of a neutron leads to the formation of fissile 233U. This process is called nuclear fuel breeding.