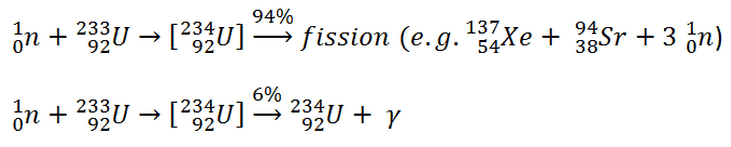Uranium 233
Uranium 233 is not a naturally occurring isotope of uranium. It is a manufactured isotope and is a key fissile isotope in the thorium fuel cycle. This isotope has a half-life of 159,200 years. 233U is produced by radiative neutron capture in nuclear reactors containing thorium 232.
233U is a fissile isotope, which means 233U can undergo a fission reaction after absorbing a thermal neutron. Moreover, 233U also meets the alternative requirement that the amount (~2.48 per one fission by thermal neutron) of neutrons produced by 233U is sufficient to sustain a nuclear fission chain reaction.
For the 233U nuclei, the number of fission neutrons produced per absorption in the fuel (the reproduction factor – η) is greater than 2.0 over a wide range of thermal neutron spectrum, unlike 235U and 239Pu. Therefore breeding can be obtained with fast, epithermal, or thermal spectra.
233U decays via alpha decay into 229Th. The decay chain of 233U itself is in the neptunium series.
232Th is the predominant isotope of natural thorium. If this fertile material is loaded in the nuclear reactor, the nuclei of 232Th absorb a neutron and become nuclei of 233Th. The half-life of 233Th is approximately 21.8 minutes. 233Th decays (negative beta decay) to 233Pa (protactinium), whose half-life is 26.97 days. 233Pa decays (negative beta decay) to 233U, which is a very good fissile material. On the other hand, proposed reactor designs must physically isolate the protactinium from further neutron capture before beta decay occurs.
Uranium 233 Fission
Uranium 233 is a very good fissile isotope, and its fission cross-section for thermal neutrons is about 531 barns (for 0.0253 eV neutron). For fast neutrons, its fission cross-section is on the order of barns. Most absorption reactions result in fission reaction, but a minority results in radiative capture forming 234U. The cross-section for radiative capture for thermal neutrons is about 45 barns (for 0.0253 eV neutron). Therefore about 6% of all absorption reactions result in radiative capture of neutrons. About 94% of all absorption reactions result in fission. The capture-to-fission ratio is much smaller than the other two major fissile fuels 235U and 239U.
Typically, when uranium 233 nucleus undergoes fission, the nucleus splits into two smaller nuclei (triple fission can also rarely occur), along with a few neutrons (the average is 2.48 neutrons per fission for thermal fission) and release of energy in the form of heat and gamma rays. The average of the fragment atomic mass is about 112, but very few fragments near that average are found. It is much more probable to break up into unequal fragments, and the most probable fragment masses are around mass 93 (strontium) and 137 (xenon).
Most of the fission fragments are highly unstable (radioactive) nuclei and undergo further radioactive decays to stabilize itself, therefore part of the released energy is radiated away from the reactor (See also: Reactor antineutrinos). On the other hand most of the energy released by one fission (~168MeV of total ~200MeV) appears as kinetic energy of these fission fragments. The fission fragments interact strongly with the surrounding atoms or molecules traveling at high speed, causing them to ionize. Creation of ion pairs requires energy, which is lost from the kinetic energy of the charged fission fragment causing it to decelerate. The positive ions and free electrons created by the passage of the charged fission fragment will then reunite, releasing energy in the form of heat (e.g., vibrational energy or rotational energy of atoms). This is the principle how fission fragments heat up fuel in the reactor core.
 Fission fragments after a nucleus fission. Fission fragments interact strongly with the surrounding atoms or molecules traveling at high speed, causing them to ionize.Source of nuclear data: JANIS (Java-based Nuclear Data Information Software); ENDF/B-VII.1
Fission fragments after a nucleus fission. Fission fragments interact strongly with the surrounding atoms or molecules traveling at high speed, causing them to ionize.Source of nuclear data: JANIS (Java-based Nuclear Data Information Software); ENDF/B-VII.1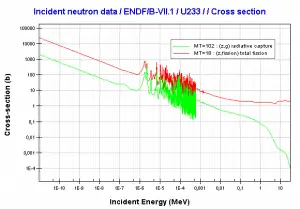 Uranium 233. Comparison of total fission cross-section and cross-section for radiative capture.
Uranium 233. Comparison of total fission cross-section and cross-section for radiative capture.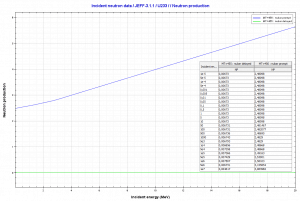 Neutron production per one fast fission of uranium 233.
Neutron production per one fast fission of uranium 233.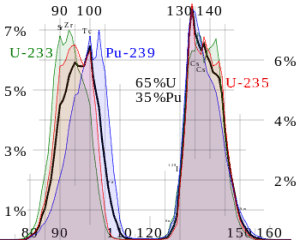 Fission fragment yield for different nuclei. The most probable fragment masses are around mass 93 (Strontium) and 137 (Xenon).
Fission fragment yield for different nuclei. The most probable fragment masses are around mass 93 (Strontium) and 137 (Xenon).