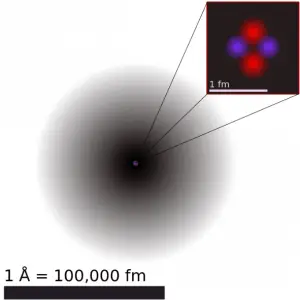
The volume of an atom is about 15 orders of magnitude larger than the volume of a nucleus. For uranium atom, the Van der Waals radius is about 186 pm = 1.86 ×10−10 m. The Van der Waals radius, rw, of an atom is the radius of an imaginary hard-sphere representing the distance of closest approach for another atom. The uranium atom has a volume of about 26.9 ×10−30 m3, assuming a spherical shape. But this “huge” space is occupied primarily by electrons because the nucleus occupies only about 1721×10−45 m3 of space. These electrons together weigh only a fraction (let say 0.05%) of the entire atom.
It may seem that the space and the matter are empty, but it is not. Due to the quantum nature of electrons, the electrons are not pointing particles, and they are smeared out over the whole atom. The classical description cannot be used to describe things on the atomic scale. On the atomic scale, physicists have found that quantum mechanics describes things very well on that scale. Particle locations in quantum mechanics are not at an exact position. They are described by a probability density function. Therefore the space in an atom (between electrons and an atomic nucleus) is not empty. Still, it is filled by a probability density function of electrons (usually known as “electron cloud“).