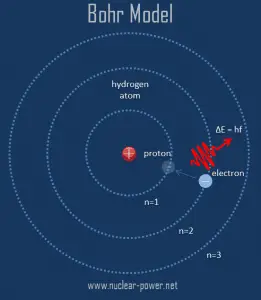
 As was written, all matter except dark matter is made of molecules, which are made of atoms. The atoms are the smallest constituents of ordinary matter, which can be divided without releasing electrically charged particles. The atoms consist of two parts. An atomic nucleus and an electron cloud, and the electrons are spinning around the atomic nucleus. The nucleus itself is generally made of protons and neutrons.
As was written, all matter except dark matter is made of molecules, which are made of atoms. The atoms are the smallest constituents of ordinary matter, which can be divided without releasing electrically charged particles. The atoms consist of two parts. An atomic nucleus and an electron cloud, and the electrons are spinning around the atomic nucleus. The nucleus itself is generally made of protons and neutrons.
This model is today quite clear and proven. But the path to this model was complex and full of many surprising findings. A scientific theory that deals with the nature of matter is known as the atomic theory. The atomic theory began as a philosophical concept in ancient Greece and India. It must be added that atomism was one of many competing theories on the nature of matter. These ancient philosophers speculated that the earth was composed of different combinations of basic substances or elements.
The word “atom” was coined by the ancient Greek philosophers Leucippus, Epicurus, and Democritus. They supposed that the properties of materials were determined by their atoms’ different shapes and forms. This word comes from the Ancient Greek adjective atomos, meaning “indivisible.”
Other ancient Greek philosophers (Empedocles, Heraclitus, and Aristotle) considered these basic elements earth, air, water, and fire. According to their philosophy, lead differs from gold only in the proportions of these four elements it contains. So the problem of converting lead into gold is just a matter of correctly blending the right combination of elements. Unlike the atomism of Democritus and Epicurus, the Aristotelian “natural minimum” was not conceptualized as physically indivisible.
Modem ideas about the structure of matter originated in the 17th century. In 1661 the English chemist Robert Boyle laid down the modern criterion of an element and published the modern criterion an element:
“a basic substance that cannot be broken down into any simpler substance after it is isolated from a compound, but can be combined with other elements to form compounds.”
To date, 105 different elements have been confirmed to exist. Of the 105 confirmed elements, 90 exist in nature, and 15 are manufactured.