Nuclear Reactors
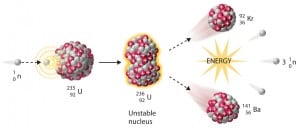
A nuclear reactor is a key device of nuclear power plants, nuclear research facilities, or nuclear-propelled ships. The main purpose of the nuclear reactor is to initiate and control a sustained nuclear chain reaction. The nuclear chain reaction is initiated, sustained, and controlled just via the free neutrons. The term chain means that one single nuclear reaction (neutron-induced fission) causes an average of one or more subsequent nuclear reactions, thus leading to the possibility of a self-propagating series of these reactions. The “one or more” is the key parameter of reactor physics. To raise or lower the power, the amount of reactions, respectively the amount of the free neutrons in the nuclear core, must be changed (using the control rods).
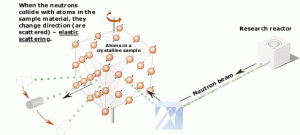
Neutron diffraction
Source: www.psi.ch
Neutron diffraction experiments use an elastic neutron scattering to determine the atomic (or magnetic) structure. The neutron diffraction is based on the fact that thermal or cold neutrons have wavelengths similar to atomic spacings. An examined sample (crystalline solids, gasses, liquids, or amorphous materials) must be placed in a neutron beam of thermal (0.025 eV) or cold (neutrons in thermal equilibrium with very cold surroundings such as liquid deuterium) neutrons to obtain a diffraction pattern that provides information about the structure of the examined material. The neutron diffraction experiments are similar to X-ray diffraction experiments, but neutrons interact with matter differently. Photons (X-rays) interact primarily with the electrons surrounding (atomic electron cloud) a nucleus, but neutrons interact only with nuclei. Neither the electrons surrounding (atomic electron cloud) a nucleus nor the electric field caused by a positively charged nucleus affect a neutron’s flight. Due to their different properties, both methods (neutron diffraction and X-ray diffraction) can provide complementary information about the material’s structure.
Applications in Medicine
Medical applications of neutrons began soon after the discovery of this particle in 1932. Neutrons are highly penetrating matter and ionizing to be used in medical therapies such as radiation therapy or boron capture therapy. Unfortunately, when they are absorbed in matter, neutrons active the matter and leave the matter (target area) radioactive.
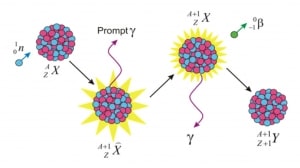
Neutron activation analysis
Source: www.naa-online.net
Neutron activation analysis is a method for determining the composition of examined material. This method was discovered in 1936 and stood at the forefront of methods used to analyze major, minor, trace, and rare elements quantitatively. This method is based on neutron activation, where an analyzed sample is first irradiated with neutrons to produce specific radionuclides. The radioactive decay of these produced radionuclides is specific for each element (nuclide). Each nuclide emits the characteristic gamma rays, measured using gamma spectroscopy, where gamma rays detected at particular energy indicate a specific radionuclide and determine concentrations of the elements. The main advantage of this method is that neutrons do not destroy the sample. This method can also be used to determine the enrichment of nuclear material.