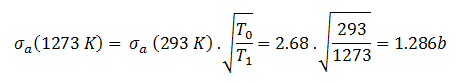Thermal Neutron – Reactors
Free neutrons can be classified according to their kinetic energy. This energy is usually given in electron volts (eV). The term temperature can also describe this energy representing thermal equilibrium between a neutron and a medium with a certain temperature.
Thermal Neutrons are neutrons in thermal equilibrium with a surrounding temperature of 290K (17 °C or 62 °F). Most probable energy at 17°C (62°F) for Maxwellian distribution is 0.025 eV (~2 km/s). This part of the neutron’s energy spectrum constitutes the most important spectrum in thermal reactors.
Thermal neutrons have a different and often much larger effective neutron absorption cross-section (fission or radiative capture) for a given nuclide than fast neutrons. Therefore the criticality of a thermal reactor can be achieved with a much lower enrichment of nuclear fuel.
Moreover, thermal neutrons are in the 1/v region, and the cross-section behaves according to the 1/v Law. In this region, the absorption cross-section increases as the neutron’s velocity (kinetic energy) decrease. Therefore the 1/v Law can be used to determine a shift in absorption cross-section if the neutron is in equilibrium with a surrounding medium. This phenomenon is because the nuclear force between the target nucleus and the neutron has a longer time to interact.
See also: Compound Nucleus Reactions.
Example of cross-sections in 1/v region:
The absorbtion cross-section for 238U at 20°C = 293K (~0.0253 eV) is:
.
The absorption cross-section for 238U at 1000°C = 1273K is equal to:
This cross-section reduction is caused only due to the shift of temperature of surrounding medium.
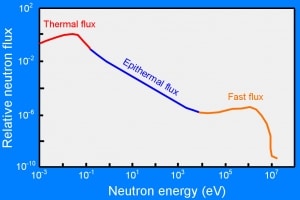 Distribution of kinetic energies of neutrons in the thermal reactor. The fission neutrons (fast flux) are immediately slowed down to the thermal energies via a process called neutron moderation.
Distribution of kinetic energies of neutrons in the thermal reactor. The fission neutrons (fast flux) are immediately slowed down to the thermal energies via a process called neutron moderation.Source: serc.carleton.edu
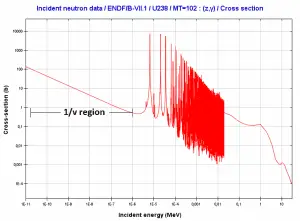 For thermal neutrons (in 1/v region), absorption cross sections increases as the velocity (kinetic energy) of the neutron decreases.
For thermal neutrons (in 1/v region), absorption cross sections increases as the velocity (kinetic energy) of the neutron decreases.Source: JANIS 4.0
Thermal Neutron – Diffraction
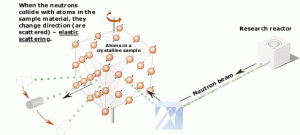
Thermal neutrons are also widely used in neutron diffraction experiments. Neutron diffraction experiments use an elastic neutron scattering to determine the atomic (or magnetic) structure of a material. The neutron diffraction is based on the fact that thermal or cold neutrons have wavelengths similar to atomic spacings.
An examined sample (crystalline solids, gasses, liquids, or amorphous materials) must be placed in a neutron beam of thermal (0.025 eV) or cold (neutrons in thermal equilibrium with very cold surroundings such as liquid deuterium) neutrons to obtain a diffraction pattern that provides information about the structure of the examined material. The neutron diffraction experiments are similar to X-ray diffraction experiments, but neutrons interact with matter differently. Photons (X-rays) interact primarily with the electrons surrounding (atomic electron cloud) a nucleus, but neutrons interact only with nuclei. Neither the electrons surrounding (atomic electron cloud) a nucleus nor the electric field caused by a positively charged nucleus affect a neutron’s flight. Due to their different properties, both methods together (neutron diffraction and X-ray diffraction) can provide complementary information about the material’s structure.