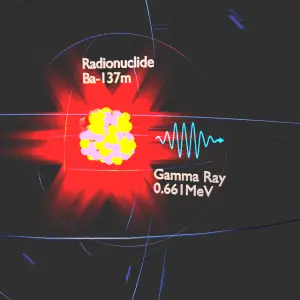
Gamma rays, also known as gamma radiation, refers to electromagnetic radiation (no rest mass, no charge) of a very high energies. Gamma rays are high-energy photons with very short wavelengths and thus very high frequency. Since the gamma rays are in substance only very high-energy photons, they are very penetrating matter and are thus biologically hazardous. Gamma rays can travel thousands of feet in the air and can easily pass through the human body. Gamma rays are emitted by unstable nuclei in their transition from a high-energy state to a lower state known as gamma decay. In most practical laboratory sources, the excited nuclear states are created in the decay of a parent radionuclide. Therefore a gamma decay typically accompanies other forms of decay, such as alpha or beta decay. Radiation and also gamma rays are all around us. In, around, and above the world we live in. It is a part of our natural world that has been here since the birth of our planet. Natural sources of gamma rays on Earth are, among other things, gamma rays from naturally occurring radionuclides, particularly potassium-40. Potassium-40 is a radioactive isotope of potassium that has a very long half-life of 1.251×109 years (comparable to the age of Earth). This isotope can be found in soil, water also in meat and bananas. This is not the only example of a natural source of gamma rays.