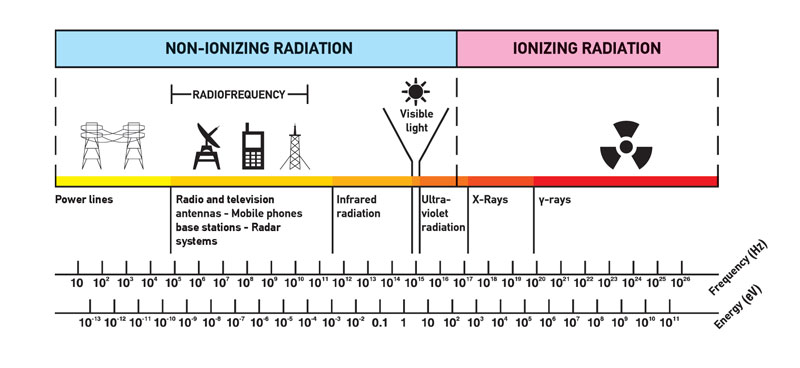
Most people connect the term radiation only with ionizing radiation, but it is incorrect. Radiation is all around us. In, around, and above the world we live in. It is a natural energy force that surrounds us. It is a part of our natural world that has been here since the birth of our planet. We should distinguish between:
- Non-ionizing radiation. The kinetic energy of particles (photons, electrons, etc.) of non-ionizing radiation is too small to produce charged ions when passing through matter. The particles (photons) have only sufficient energy to change the rotational, vibrational, or electronic valence configurations of target molecules and atoms. Sunlight, radio waves, and cell phone signals are examples of non-ionizing (photon) radiation. However, it can still cause harm, like when you get a sunburn.
- Ionizing radiation. The kinetic energy of particles (photons, electrons, etc.) of ionizing radiation is sufficient, and the particle can ionize (to form ions by losing electrons) and target atoms to form ions. Ionizing radiation can knock electrons from an atom.
The boundary is not sharply defined since different molecules and atoms ionize at different energies, which is typical for electromagnetic waves. Among electromagnetic waves belong in the order of increasing frequency (energy) and decreasing wavelength: radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays, and gamma rays. Gamma rays, X-rays, and the higher ultraviolet part of the spectrum are ionizing, whereas the lower ultraviolet, visible light (including laser light), infrared, microwaves, and radio waves are considered non-ionizing radiation.
Non-ionizing radiation refers to less energetic radiation, but it does not mean it cannot be harmful. One form of non-ionizing radiation is ultraviolet, or UV, radiation, which you may be familiar with – we use sunscreen on Earth to shield ourselves from harmful UV rays. It must be noted that UV is also responsible for forming bone-strengthening vitamin D in most land vertebrates, including humans (specifically, UVB). The UV spectrum thus has effects both beneficial and harmful to human health.
In every case, the intensity of non-ionizing radiation also matters. Consider heat from a fire. If you are too close, the intensity of heat radiation is high, and you can get burned. If you are at the right distance, you can withstand it without any problems, and it is comfortable. If you are too far from a heat source, the insufficiency of heat can also hurt you. In a certain sense, this analogy can be applied to radiation also from radiation sources.