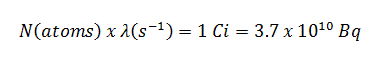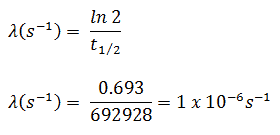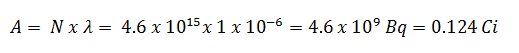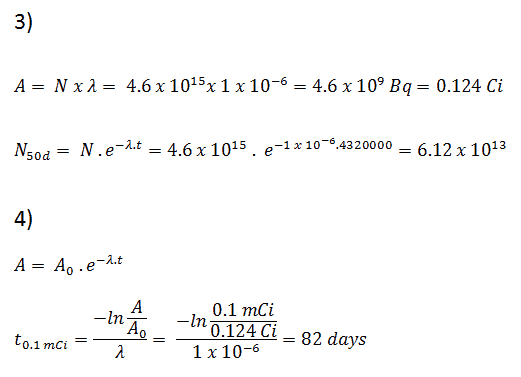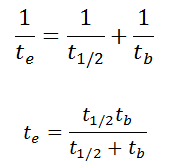 One of the most useful terms for estimating how quickly a nuclide will decay is the radioactive half-life (t1/2). The half-life is defined as the amount of time it takes for a given isotope to lose half of its radioactivity. As was written, radioactive decay is a random process at the level of single atoms. According to quantum theory, predicting when a particular atom will decay is impossible. In other words, a nucleus of a radionuclide has no “memory,” and a nucleus does not “age” over time. Thus, the probability of its breaking down does not increase with time but stays constant no matter how long the nucleus has existed.
One of the most useful terms for estimating how quickly a nuclide will decay is the radioactive half-life (t1/2). The half-life is defined as the amount of time it takes for a given isotope to lose half of its radioactivity. As was written, radioactive decay is a random process at the level of single atoms. According to quantum theory, predicting when a particular atom will decay is impossible. In other words, a nucleus of a radionuclide has no “memory,” and a nucleus does not “age” over time. Thus, the probability of its breaking down does not increase with time but stays constant no matter how long the nucleus has existed.
Therefore, the rate of nuclear decay can also be measured in terms of half-lives. Each radionuclide has its own particular half-life that never changes, regardless of the quantity or form of the material (i.e., solid, liquid, gas, element, or compound) or its history. If a radioisotope has a half-life of 14 days, half of its atoms will have decayed within 14 days. In 14 more days, half of that remaining half will decay, and so on.
Half-lives range from millionths of a second for highly radioactive fission products to billions of years for long-lived materials (such as naturally occurring uranium). After five half-lives have elapsed, only 1/32, or 3.1%, of the original number of atoms remains. After seven half-lives, only 1/128, or 0.78%, of the atoms remain. The number of atoms existing after 5 to 7 half-lives can usually be assumed to be negligible.
The fraction of the original activity remaining after succeeding half-lives is:
Activity after 1 half-life = ½ of the original
Activity after 2 half-lives = ½ x ½ = ¼ of the original
Activity after 3 half-lives = ½ x ½ x ½ = (½)3 = 1/8 of the original
Activity after 4 half-lives = (½)4 = 1/16 of the original
Activity after 5 half-lives = (½)5 = 1/32 of the original
Activity after 6 half-lives = (½)6 = 1/64 of the original
Activity after 7 half-lives = (½)7 = 1/128 of the original
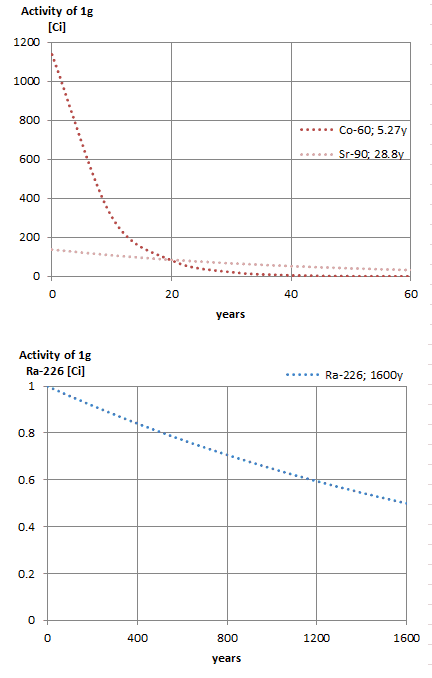 Notice that short half-lives go with large decay constants. Radioactive material with a short half-life is much more radioactive (at the time of production) but will obviously lose its radioactivity rapidly. No matter how long or short the half-life is after seven half-lives have passed, there is less than 1 percent of the initial activity remaining.
Notice that short half-lives go with large decay constants. Radioactive material with a short half-life is much more radioactive (at the time of production) but will obviously lose its radioactivity rapidly. No matter how long or short the half-life is after seven half-lives have passed, there is less than 1 percent of the initial activity remaining.
The radioactive decay law states that the probability per unit time that a nucleus will decay is a constant, independent of time. This constant is called the decay constant and is denoted by λ, “lambda.” This constant probability may vary greatly between different types of nuclei, leading to the many different observed decay rates. The radioactive decay of a certain number of atoms (mass) is exponential in time.
Radioactive decay law: N = N.e-λt
The rate of nuclear decay is also measured in terms of half-lives. The half-life is the amount of time it takes for a given isotope to lose half of its radioactivity. If a radioisotope has a half-life of 14 days, half of its atoms will have decayed within 14 days. In 14 more days, half of that remaining half will decay, and so on. Half-lives range from millionths of a second for highly radioactive fission products to billions of years for long-lived materials (such as naturally occurring uranium). Notice that short half-lives go with large decay constants. Radioactive material with a short half-life is much more radioactive (at the time of production) but will obviously lose its radioactivity rapidly. No matter how long or short the half-life is after seven half-lives have passed, there is less than 1 percent of the initial activity remaining.
The radioactive decay law can also be derived for activity calculations or mass of radioactive material calculations:
(Number of nuclei) N = N.e-λt (Activity) A = A.e-λt (Mass) m = m.e-λt
where N (number of particles) is the total number of particles in the sample, A (total activity) is the number of decays per unit time of a radioactive sample, and m is the mass of remaining radioactive material.
Half-Life and Decay Constant
In radioactivity calculations, one of two parameters (decay constant or half-life), which characterize the decay rate, must be known. There is a relation between the half-life (t1/2) and the decay constant λ. The relationship can be derived from the decay law by setting N = ½ No. This gives:
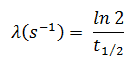 where ln 2 (the natural log of 2) equals 0.693. If the decay constant (λ) is given, it is easy to calculate the half-life and vice-versa.
where ln 2 (the natural log of 2) equals 0.693. If the decay constant (λ) is given, it is easy to calculate the half-life and vice-versa.
Half-Life and Radioactivity
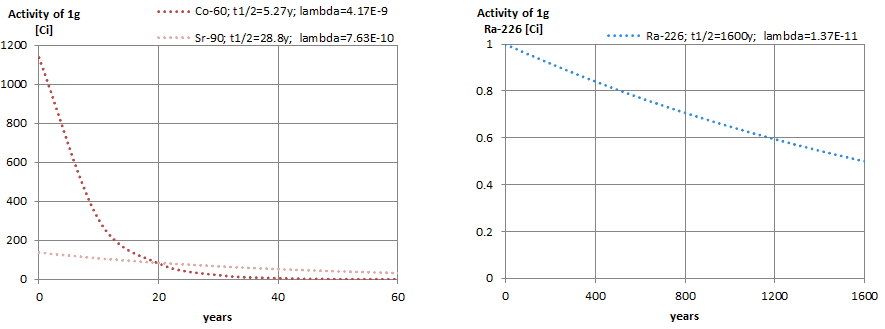
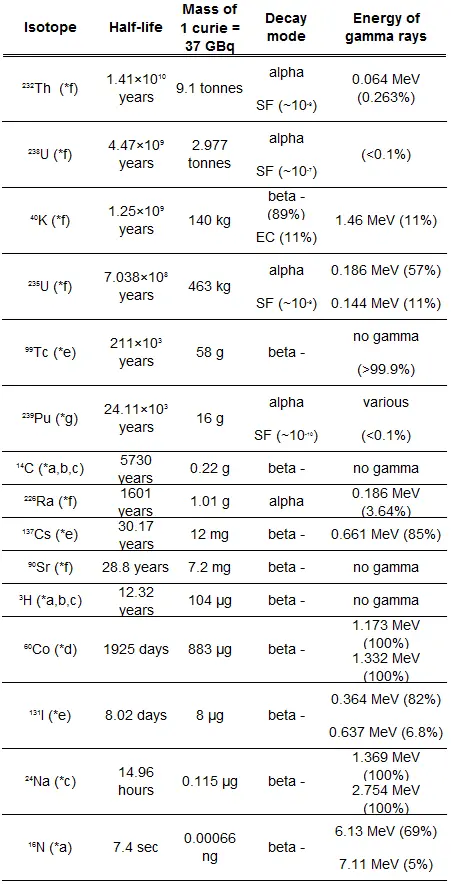
The relationship between half-life and the amount of a radionuclide required to give an activity of one curie is shown in the figure. This amount of material can be calculated using λ, which is the decay constant of certain nuclide:
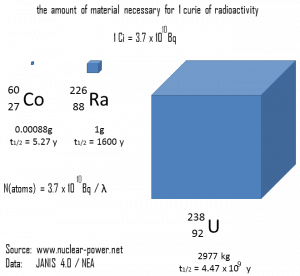 The following figure illustrates the amount of material necessary for 1 curie of radioactivity. Obviously, the longer the half-life, the greater the quantity of radionuclide needed to produce the same activity. Of course, the longer-lived substance will remain radioactive for much longer. As can be seen, the amount of material necessary for 1 curie of radioactivity can vary from an amount too small to be seen (0.00088 gram of cobalt-60), through 1 gram of radium-226, to almost three tons of uranium-238.
The following figure illustrates the amount of material necessary for 1 curie of radioactivity. Obviously, the longer the half-life, the greater the quantity of radionuclide needed to produce the same activity. Of course, the longer-lived substance will remain radioactive for much longer. As can be seen, the amount of material necessary for 1 curie of radioactivity can vary from an amount too small to be seen (0.00088 gram of cobalt-60), through 1 gram of radium-226, to almost three tons of uranium-238.
Example – Radioactive Decay Law
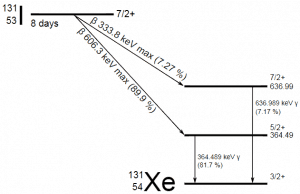 A sample of material contains 1 microgram of iodine-131. Note that iodine-131 plays a major role as a radioactive isotope present in nuclear fission products and is a major contributor to the health hazards when released into the atmosphere during an accident. Iodine-131 has a half-life of 8.02 days.
A sample of material contains 1 microgram of iodine-131. Note that iodine-131 plays a major role as a radioactive isotope present in nuclear fission products and is a major contributor to the health hazards when released into the atmosphere during an accident. Iodine-131 has a half-life of 8.02 days.
Calculate:
- The number of iodine-131 atoms is initially present.
- The activity of the iodine-131 in curies.
- The number of iodine-131 atoms will remain in 50 days.
- The time it will take for the activity to reach 0.1 mCi.
Solution:
- The number of atoms of iodine-131 can be determined using isotopic mass as below.
NI-131 = mI-131 . NA / MI-131
NI-131 = (1 μg) x (6.02×1023 nuclei/mol) / (130.91 g/mol)
NI-131 = 4.6 x 1015 nuclei
- The activity of the iodine-131 in curies can be determined using its decay constant:
The iodine-131 has a half-life of 8.02 days (692928 sec), and therefore its decay constant is:
Using this value for the decay constant, we can determine the activity of the sample:
3) and 4) The number of iodine-131 atoms that will remain in 50 days (N50d) and the time it will take for the activity to reach 0.1 mCi can be calculated using the decay law:
As can be seen, after 50 days, the number of iodine-131 atoms and thus the activity will be about 75 times lower. After 82 days, the activity will be approximately 1200 times lower. Therefore, the time of ten half-lives (factor 210 = 1024) is widely used to define residual activity.
Biological Half-Life
In general, the biological half-life is the time taken for the amount of a particular element in the body to decrease to half of its initial value due to elimination by biological processes alone, when the rate of removal is roughly exponential. The biological half-live (tbiological) can be defined as metabolites, drugs, and other substances, and it is also very important in radiation protection when considering internal exposure.
If the radiation source is inside our body, we say it is internal exposure. The intake of radioactive material can occur through various pathways such as ingesting radioactive contamination in food or liquids, inhalation of radioactive gases, or through intact or wounded skin. Most radionuclides will give you much more radiation dose if they can somehow enter your body than they would if they remained outside. The biological half-life depends on the rate at which the body normally uses a particular compound of an element. Radioactive isotopes that were ingested or taken in through other pathways will gradually be removed from the body via bowels, kidneys, respiration, and perspiration. This means that a radioactive substance can be expelled before it has had the chance to decay.
As a result, the biological half-life significantly influences the overall dose from internal contamination. If a radioactive compound with a radioactive half-life (t1/2) is cleared from the body with a biological half-life tb, the “effective” half-life (te) is given by the expression:
As can be seen, the biological mechanisms always decrease the overall dose from internal contamination. Moreover, if t1/2 is large compared to tb, the effective half-life is approximately the same as tb. For example, tritium has a biological half-life of about 10 days, while the radioactive half-life is about 12 years.
See also: Biological Half-Life
See also: Effective Half-Life