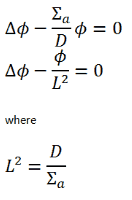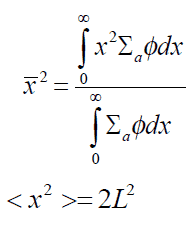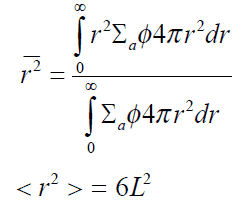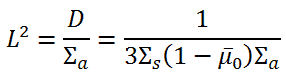L2 is equal to one-sixth of the square of the average distance (in all dimensions) between the neutron’s birth point (as a thermal neutron) and its absorption.
During the diffusion equation solution, we often meet with a very important parameter that describes the behavior of neutrons in a medium.
The solution of diffusion equation (let assume the simplest diffusion equation) usually starts by division of entire equation by diffusion coefficient:
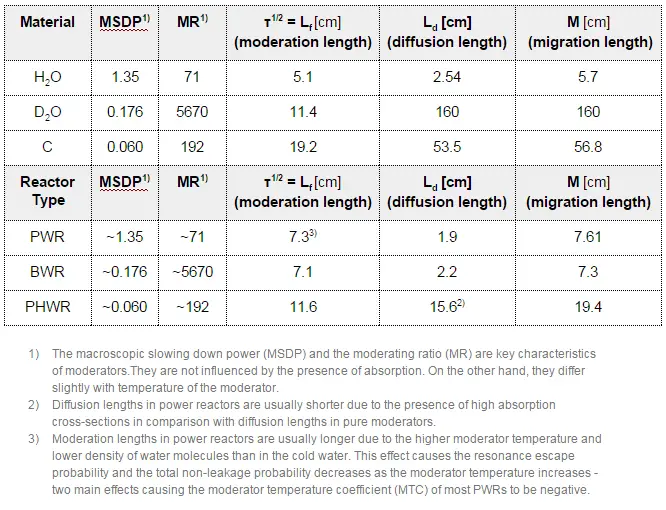
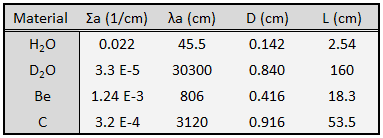
The term L2 is called the diffusion area (and L is called the diffusion length). For thermal neutrons with an energy of 0.025 eV, a few values of L are given in the table below.
Physical Meaning of the Diffusion Length
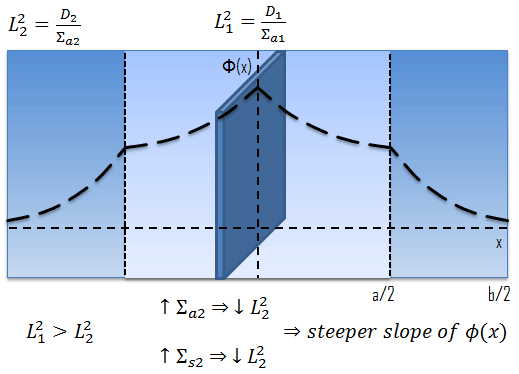
It is interesting to try to interpret the “physical” meaning of the diffusion length. The physical meaning of the diffusion length can be seen by calculating the mean square distance that a neutron travels in the one direction from the plane source to its absorption point.
It can be calculated that L2 is equal to one-half the square of the average distance (in one dimension) between the neutron’s birth point and its absorption.
If we consider a point source of neutrons, the physical meaning of the diffusion length can be seen again by calculating the mean square distance that a neutron travels from the source to its absorption point.
It can be calculated that:
L2 is equal to one-sixth of the square of the average distance (in all dimensions) between the neutron’s birth point (as a thermal neutron) and its absorption.
This distance must not be confused with the average distance traveled by the neutrons. The average distance traveled by the neutrons is equal to the mean free path for absorption λa = 1/Σa and is much larger than the distance measured in a straight line. This is because neutrons in medium undergo many collisions and they follow a very zig-zag path through medium.
Operational changes that affect the diffusion length
The diffusion length is an important parameter in thermal reactors, and its magnitude can be changed during reactor operation. Since the diffusion length is dependent on the diffusion coefficient (~Σs) and Σa, we will study the impacts of operational changes on these parameters. For illustration, here are some examples of these operational changes and their impacts on L2 that may take place in PWRs.
Change in the moderator and fuel temperature
The diffusion length, L2, is sensitive, especially on the change in the moderator temperature. Since diffusion in heterogeneous reactors occurs especially in the moderator, the change in the moderator temperature dominates over the change in the fuel temperature.
In short, as the moderator temperature increases, the diffusion length also increases.
The moderator temperature influences all macroscopic cross-sections (e.g.,, Σs=σs.NH2O), especially due to the thermal expansion of water, which results in a decrease in the atomic number density. The microscopic cross-sections also slightly change with temperature, but not so much in the thermal spectrum.
For the diffusion length, there are two effects. Both processes have the same direction and together cause the increase in the diffusion lengths as the temperature increases. Since the diffusion length significantly influences the thermal non-leakage probability, it is important in reactor dynamics (it influences moderator temperature feedback).
- Macroscopic cross-sections for elastic scattering reaction Σs=σs.NH2O, which significantly changes due to the thermal expansion of water. As the temperature of the core increases, the diffusion coefficient (D = 1/3.Σtr) increases.
- The macroscopic cross-section for neutron absorption also decreases as the moderator temperature increases. This is especially due to the thermal expansion of water and the changes in the microscopic cross-section (σa) for neutron absorption. As the temperature of the core increases, the absorption cross-section decreases.
Change in the boron concentration
The concentration of boric acid diluted in the primary coolant influences the diffusion length. For example, an increase in the concentration of boric acid (chemical shim) causes an addition of new absorbing material into the core and this causes an increase in the macroscopic absorption cross-section and this, in turn, causes a decrease in the diffusion length.